 
Nevertheless, the general meaning of the energy-time principle is that a quantum state that exists for only a short time cannot have a definite energy. (C) The revolutionar meaniny ogf quantu theorm foy r theolog. Whether ontological or epistemological, however, uncertainty appears to be at th e hear ot f every interpretatio of microworln realityd. But the act of measurement involves interacting with the electron, which will cause it to change its momentum, increasing your uncertainty about that value.For technical reasons beyond this discussion. Theology and the Heisenberg Uncertainty Principle 131 uncertainty is a principle of genuine indeterminacy an d not merely a principle of ignorance. Instead, they are more like clouds of probabilities, with a varying chance of appearing in a certain place, which gives them a wave-like nature.Īs you repeatedly measure an electron, you can build up certainty about its position. The Heisenberg Uncertainty Principle is a fundamental theory in quantum mechanics that defines why a scientist cannot measure multiple quantum variables simultaneously. 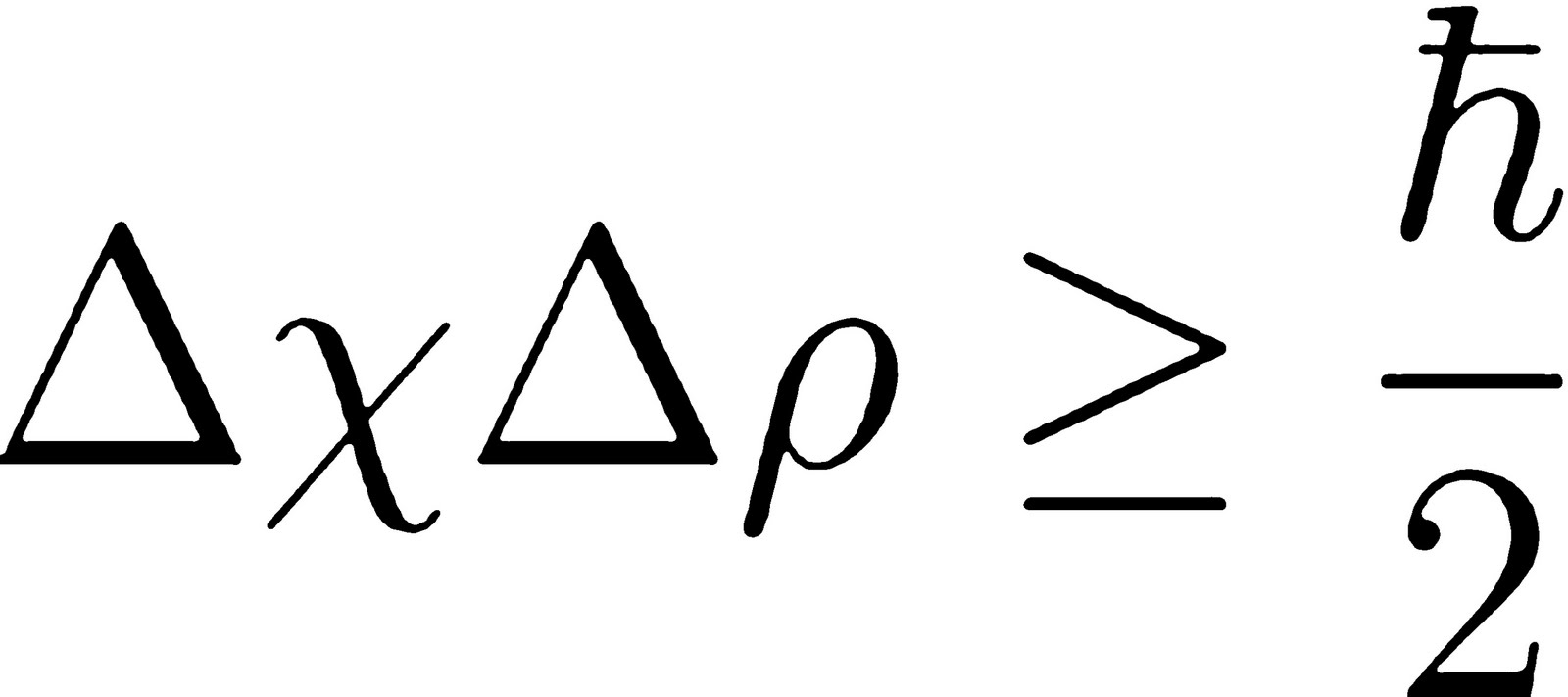
Particles governed by quantum physics, such as the electron, don’t really exist in a specific place. Heisenberg uncertainty principle synonyms, Heisenberg uncertainty principle pronunciation, Heisenberg uncertainty principle translation, English dictionary definition of Heisenberg uncertainty principle. The Heisenberg uncertainty principle says that we cannot know both the position and the momentum of a particle at once. It’s a concept that makes no sense on a human scale, but that’s quantum mechanics for you. The uncertainty principle says that it is not possible to precisely and simultaneously measure both the velocity and position of a subatomic particle like an electron. The two error sizes, when multiplied together, will always exceed a certain value. 
It states that any effort to reduce the error in your measurement of a particle’s momentum (that is, its mass multiplied by its velocity) will increase the error in your measurement of its position. Here q is the uncertainty in the position of the particle (in metres), v is the. Explore the Heisenberg uncertainty principle by calculating uncertainty in position given the uncertainty in momentum for Bohr model of hydrogen. This bizarre trade-off arises from Heisenberg’s uncertainty principle, which was first laid out by German physicist Werner Heisenberg in 1927. For the example given earlier, Heisenberg’s principle can be precisely stated as: (1) q x v > /m. The Heisenberg uncertainty principle states that there is a limit to how precisely certain pairs of physical properties of a particle can be known simultaneously. For quantum particles, this is a reality: the better you know a particle’s speed, the less certain you are of its position. If you are using the velocity, you will also have to define the mass of the object: we will use these two quantities to calculate the momentum uncertainty. Chad Orzel navigates this complex concept of quantum physics. Why not Because everything in the universe behaves like both a particle and a wave at the same time. Imagine driving a car fitted with a GPS navigation system that glitches every time you look at the speedometer. 2.1 Heisenberg’s road to the uncertainty relations 2.2 Heisenberg’s argument 2.3 The interpretation of Heisenberg’s uncertainty relations 2. To use our Heisenbergs uncertainty principle calculator, you have to know the values of the uncertainty of one of three possible quantities: The momentum The position or. The Heisenberg Uncertainty Principle states that you can never simultaneously know the exact position and the exact speed of an object. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
